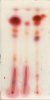
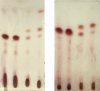
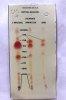
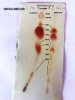
I did try ordering from one supplier (Santa Cruz Biotechnology) and they emailed saying they couldn’t ship anything to a residential address. I’ve since checked with about 10 different companies and everyone advertises that they won’t ship to residential addresses or that they “inform law enforcement of any unusual purchases.” To my knowledge it’s not illegal to posses these chemicals, but I don’t want to work with any company that threatens its customers before even ordering.
I have been using the tiny sample of Fast Blue BB that came with a test kit I bought from cannalyticssupply.com. They are selling a replacement vial that holds 150mg of Fast Blue BB for $15, which I’ve since reluctantly purchased. That’s $0.10/mg from the test kit vendor versus the $0.005/mg from Santa Barbara Biotechnology. I’m still looking for a larger quantity to lower the per unit cost.
I have been using the tiny sample of Fast Blue BB that came with a test kit I bought from cannalyticssupply.com. They are selling a replacement vial that holds 150mg of Fast Blue BB for $15, which I’ve since reluctantly purchased. That’s $0.10/mg from the test kit vendor versus the $0.005/mg from Santa Barbara Biotechnology. I’m still looking for a larger quantity to lower the per unit cost.